Students can refer to the following Periodic Classification of Elements MCQ Questions for Class 10 with Answers provided below based on the latest curriculum and examination pattern issued by CBSE and NCERT. Our teachers have provided here a collection of multiple choice questions for Periodic Classification of Elements Class 10 covering all topics in your textbook so that students can assess themselves on all important topics and thoroughly prepare for their exams
Periodic Classification of Elements MCQ Questions for Class 10 with Answers
We have provided below Periodic Classification of Elements MCQ Questions for Class 10 with answers which will help the students to go through the entire syllabus and practice multiple choice questions provided here with solutions. As Periodic Classification of Elements MCQs for Class 10 Science pdf download can be really scoring for students, you should go through all problems provided below so that you are able to get more marks in your exams.
Question.. The longest and the shortest periods are:
a. 1&6
b. 2&6
c. 6 &1
d. 1&7
Answer
B
Question.. The number of elements present in the 2nd, 3rd, 4th and 5th periods of the modern periodic table are:
a. 2,8,8,18
b. 8,8,18,32
c. 8,8,18, 18
d. 8,18,18,32
Answer
B
Question.. The pairs of elements with the following atomic numbers have the same chemical properties:
a. 13 & 12
b. 3 &11
c. 4&24
d. 2 &1
Answer
B
Question.. The period that contains only gaseous elements are:
a. 1
b. 2
c. 3
d. 4
Answer
A
Question.. Elements with atomic number 15 and mass number 31 is present in:
a. Group 5 and period 4
b. Group5 and period 3
c. Group15 and period 3
d. Group15 and period 4
Answer
C
Question.. Which of the following will form acidic oxide? An element with atomic number:
a. 7
b. 11
c. 21
d. 19
Answer
A
Question.. Which amongst the following represents the correct order of decreasing metallic character of elements Na, Si, Cl, Mg, Al
a. Cl> Si> Al> Mg> Na
b. Na> Mg> Al> Si> Cl
c. Na> Si> Mg> Al> Cl
d. Al> Na> Si> Cl> Mg
Answer
B
Question.. Which of the following are characteristics of isotopes of an element?
a. Isotopes of an element have same atomic masses
b. Isotopes of an element have same atomic number
c. Isotopes of an element show same physical properties
d. Isotopes of an element have same chemical properties
i. A, c, d
ii. B, c, d
iii. B and c
iv. B and d
Answer
C
Question.. Where would you locate an element with electronic configuration 2, 8,7 in the modern periodic table? B
a. Group 7 and period 2
b. Group7 and period 3
c. Group 17 and period 3
d. Group17 and period 3
Answer
Question.. Which of the given elements A, B , C, D and E with atomic numbers 2, 4, 8, 10 and 18 respectively belong to the same period?
a. A, B, C
b. B,C,D
c. A,D,E
d. B,D,E
Answer
B
Question.. Which of the following hydroxides are most basic:
a. Be(OH)2
b. Mg(OH)2
c. Ca(OH)2
d. Ba(OH)2
Answer
D
Question.. An element has 13 protons. The group and period to which it belongs:
a. 3rd period and 13th group
b. 2nd period and 13th group
c. 3rd period and 3rd group
d. 2nd period and 3rd group
Answer
A
Question.. Which of the following is the correct order of size:
a. Cl< F< Br< I
b. F< Cl< Br< I
c. I<Br< Cl< F
d. Br< I< Cl< F
Answer
B
Question.. The lightest metal is :
a. Li
b. Na
c. K
d. Mg
Answer
A
Question.. Which of the following has most non metallic character:
a. N
b. C
c. O
d. F
Answer
D
Question.. Which of the following is the correct order of size:
a. I+ >I- > I
b. I- >I > I+
c. I >I+ > Id.
I >I- > I+
Answer
Question. Which of the following elements would accept an electron readily:
a. F
b. Cl
c. Br
d. I
Answer
b
Question.. The most metallic element in the fourth period is:
a. Ca
b. K
c. S
d. P
Answer
B
Question.. Which of the following elements would lose an electron easily:
a. K b. Na
c. Ca
d. Mg
Answer
B
Question. Cl, Br I, if this is a Dobereiner’s triad and the atomic masses of Cl and I are 35.5 and 127 respectively the atomic mass of Br is
(1) 162.5
(2) 91.5
(3) 81.25
(4) 45.625
Answer
3
Question. Which of the following hydroxides is most basic ?
(1) Be(OH)2
(2) Ba(OH)2
(3) Ca(OH)2
(4) Mg(OH)2
Answer
2
Question. Which of the following elements will form acidic oxide ?
(1) Sodium
(2) Magnesium
(3) Aluminium
(4) Sulphur
Answer
4
Question. According to Mendeleev’s periodic law, thev properties of elements are a periodic function of their
(1) Atomic number
(2) Atomic masses
(3) Atomic volumes
(4) Atomic sizes
Answer
2
Question. The alkali metals are in which group of the periodic table ?
(1) Group 1
(2) Group 2
(3) Group 3
(4) Group 4
Answer
1
Question. “The properties of the elements are periodic function of their atomic numbers”. The statement was given by
(1) N. Bohr
(2) J.W. Dobereiner
(3) D.I. Mendeleev
(4) H.G.J. Moseley
Answer
4
Question. In the periodic table, the metallic character of elements
(1) decreases from left to right and down the group
(2) decreases from left to right and increases down the group
(3) increases from left to right and down the group
(4) increases from left to right and decreases down the group
Answer
2
Question. Which pair of elements of the following sets is likely to have similar chemical behaviour ?
(1) Sodium and aluminium
(2) Argon and potassium
(3) Boron and germanium
(4) Nitrogen and phosphorus
Answer
4
Question. Gradual addition of electronic shells in the noble gases causes a decrease in their
(1) Ionisation energy
(2) Atomic radius
(3) Boiling point
(4) Density
Answer
1
Question. Which of the following elements has the least nonmetallic character ?
(1) Fluorine
(2) Chlorine
(3) Bromine
(4) Iodine
Answer
4
Question. Why are the elements lithium, sodium and potassium called alkali metals ?
(1) Because they reacts with water to form alkali
(2) Because they form acidic oxides
(3) Because they are present in first group
(4) Because they are less reactive in nature
Answer
1
Question. Which of the following oxides is amphoteric in nature ?
(1) CaO
(2) CO2
(3) SiO2
(4) SnO2
Answer
4
Case study questions
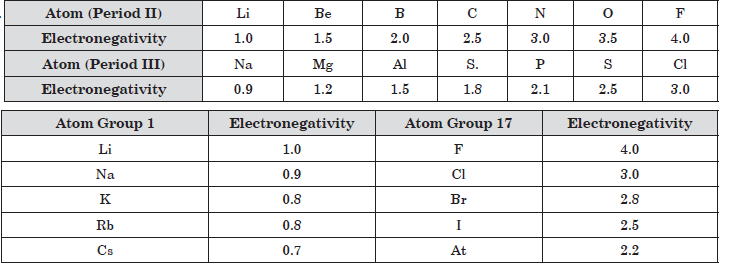
Question. How electronegativity varies in a period?
(a) Increases from left to right
(b) Decreases from left to right
(c) First increases then decreases
(d) Vary independently
Answer
A
Question. What happens to tendency to gain electron in a period?
(a) Increases,
(b) Decreases,
(c) Remaining same,
(d) First increases then decreases.
Answer
A
Question. How electronegativity varies in a period?
(a) Increases down the group
(b) Decreasing down the group
(c) First increases then decreases down the group
(d) Vary independently
Answer
B
Question. Which element has highest electronegativity?
(a) C
(b) N
(c) O
(d) F
Answer
D
Question. Which of the following has least electronegativity?
(a) Li,
(b) Be,
(c) O,
(d) N
Answer
A
Modern periodic table has 18 vertical columns known as groups and 7 horizontal rows known as periods. First period contains 2 elements second and third period contain 8 elements. 4th and 5th period contains 18 elements and 6th and 7th period contains 32 elements. The graph is plotted between atomic number and atomic radius of group 17 and group 1 elements.
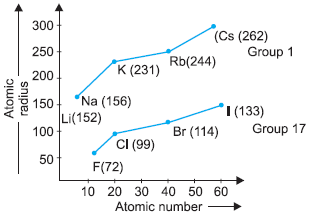
Question. Which group elements will gain electrons to form negative ions?
(a) Group 1
(b) Group 2
(c) Group 17
(d) Group 18
Answer
C
Question. Atomic size decreases from left to right in a period because
(a) Effective nuclear charge increases
(b) Number of shells remains the same
(c) Force of attraction between the nucleus and valence electrons increases
(d) All of these
Answer
D
Question. Which element in group 17 has smallest size?
(a) Flourine
(b) Bromine
(c) Chlorine
(d) Iodine
Answer
A
Question. Which group elements will have largest atomic size?
(a) Group 1
(b) Group 2
(c) Group 3
(d) Group 18
Answer
A
Question. What happens to atomic radii in a group from top to bottom?
(a) Increases
(b) Decreases
(c) First decreases then increases
(d) Number of shells remains the same
Answer
A
Around the year 1800, only 30 elements were known. Dobereiner in 1817 and Newlands in 1866 tried to arrange the then known elements and framed laws which were rejected by the scientists. Even after the rejection of the proposed laws, many scientists continued to search for a pattern that correlated the properties of elements with their atomic masses.
The main credit for classifying elements goes to Mendeleev for his most important contribution to the early development of a Periodic table of elements wherein he arranged the elements on the basis of their
fundamental property, the atomic mass and also on the similarity of chemical properties. The formulae of their hydrides and oxides were treated as basic criteria for the classification of the elements.
However, Mendeleev’s classification also had some limitations as it could not assign the position to isotopes. He also left some gaps in the periodic table.
Question. Why did Mendeleev leave some gaps in the Periodic table?
(a) For elements to be discovered
(b) For isotopes
(c) For isobars
(d) None of these
Answer
A
Question. According to Mendeleev’s Periodic Law properties of elements are periodic function of
(a) atomic mass
(b) atomic number
(c) number of protons
(d) number of electrons
Answer
A
Question. If the letter ‘R’ was used to represent any of the elements in the group, then the hydride and oxide of carbon would respectively be represented as
(a) RH4, RO
(b) RH4, RO2
(c) RH2, RO2
(d) RH2, RO
Answer
B
Question. How many groups and periods are there in Mandeleev’s periodic table?
(a) 6 group, 8 period
(b) 18 group, 7 period
(c) 7 group, 18 period
(d) 8 group, 6 period
Answer
D
Question. Isotopes are:
(a) Atoms of an element with similar chemical properties but different atomic masses.
(b) Atoms of different elements with similar chemical properties but different atomic masses.
(c) Atoms of an element with different chemical properties but same atomic masses.
(d) Atoms of different elements with different chemical properties but same atomic masses.
Answer
A
Assertion-reason Type Questions :
For question numbers 1 to 2 two statements are given‑one labeled as Assertion (A) and the other labeled
Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below:
(a) Both ‘A’ and ‘R’ are true and ‘R’ is correct explanation of the assertion.
(b) Both ‘A’ and ‘R’ are true but ‘R’ is not correct explanation of the assertion.
(c) ‘A’ is true but ‘R’ is false.
(d) ‘A’ is false but ‘R’ is true.
Question. Assertion: X with atomic number 13 is a metal.
Reason: It belongs to group 13 and 3rd period.
Answer
B
Question. Assertion: Carbon is a metalloid.
Reason: Carbon forms CO2 which is acidic oxide whereas CO is neutral oxide.
Answer
D


