Please refer to Class 12 Chemistry Sample Paper With Solutions Set B provided below. The Sample Papers for Class 12 Chemistry have been prepared based on the latest pattern issued by CBSE. Students should practice these guess papers for class 12 Chemistry to gain more practice and get better marks in examinations. The Sample Papers for Chemistry Standard 12 will help you to understand the type of questions which can be asked in upcoming examinations.
Sample Paper for Class 12 Chemistry With Solutions Set B
Topic-1
Conductance in Electrolytic Solutions, Specific and Molar Conductivity, Variations of Conductivity with Concentration,Gibbs Energy, Kohlrausch’s Law
Very Short Answer-Objective Type Questions
A. Multiple Choice Questions:
Question. Which of the following statement is not correct about an inert electrode in a cell?
(a) It does not participate in the cell reaction.
(b) It provides surface either for oxidation or for reduction reaction.
(c) It provides surface for conduction of electrons.
(d) It provides surface for redox reaction.
Answer
D
Question. Which of the statements about solutions of electrolytes is not correct?
(a) Conductivity of solution depends upon size of ions.
(b) Conductivity depends upon viscosity of solution.
(c) Conductivity does not depend upon solvation of ions present in solution.
(d) Conductivity of solution increases with temperature.
Answer
C
Question. Λ0m(NH4OH) is equal to …………………

Answer
C
B. Match the following :
Question. Match the species given in Column I with those mentioned in Column II.
Column I Column II
(i) Λm (a) Scm-1
(ii) ECell (b) m-1
(iii) κ (c) S cm2mol-1
(iv) G* (d) V
Answer. Correct option :
(i) → (c)
(ii) → (d)
(iii) → (a)
(iv) → (b)
C. Answer the following:
Question. In an aqueous solution how does specific conductivity of electrolytes change with addition of water?
Answer. Conductivity decreases because number of ions per unit volume decreases.
Short Answer Type Questions
Question. The conductivity of a 0.01 M solution of acetic acid at 298 K is 1.65 x 10–4 S cm–1.
Calculate molar conductivity (Λm) of the solution.
Answer.
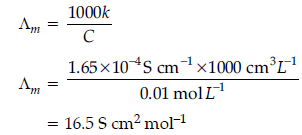
Question. Calculate the degree of dissociation (α) of acetic acid if its molar conductivity (Λm) is 39.05 S cm2 mol-1. Given L˚(H+) = 349.6 S cm2 mol-1 and Λm(CH3COO–) = 40.9 S cm2 mol-1.
Answer. Λ°CH3COOH = Λ°CH3COO- + Λ°H+
= 40.9 + 349.6 = 390.5 S cm2/mol
Now, α = Λm/Λ°m
= 39.05/390.5 = 0.1
Question. Why on dilution the Λm of CH3COOH increases drastically, while that of CH3COONa increases gradually?
Answer. In case of CH3COOH which is a weak electrolyte, the number of ions increase on dilution due to an increase in degree of dissociation resulting in drastic increase in Λm.
CH3COOH+H2O → CH3COO− +H3O+
In the case of CH3COONa which is a strong electrolyte, the number of ions remains the same but the inter-ionic attraction decreases resulting in gradual increase in Λm.
Question.Define the following terms :
(i) Molar conductivity (Lm),
(ii) Secondary batteries.
Answer. (i) Molar conductivity (Λm) : Molar conductivity is defined as the conductivity due to all the ions produced by dissolving one mole of an electrolyte in solution.
(ii) In secondary batteries, the reactions can be reversed the cell reactions imposing a higher voltage than the E.M.F. of cell (external voltage).
These batteries can be recharged by passing electric current and used again and again.
Question. The conductivity of 0.20 M solution of KCl at 298 K is 0.025 S cm–1. Calculate its molar conductivity.
Answer. Concentration of solution = 0.20 M
Conductivity = 0.025 S cm–1
Molar conductivity Λm =[Conductivity (K)×1000]/Concentration of solution

125 S cm2 mol–1
Question. Define the following terms :
(i) Fuel cell
(ii) Limiting molar conductivity ( Λmo )
Answer. (i) Galvanic cells that are designed to convert the energy of combustion of fuels (methane, methanol etc.) directly into electrical energy are called fuel cells.
(ii) When the concentration approaches zero, the molar conductivity is known as limiting molar conductivity. It is represented by Λ°m.
Question. The following curve is obtained when molar conductivity Λm) is plotted against the square root of concentration, c½ for two electrolytes A and B :

(i) How do you account for the increase in the molar conductivity of the electrolyte A on dilution ?
(ii) As seen from the graph, the value of limiting molar conductivity (Λ°m ) for electrolyte B cannot be obtained graphically. How can this value be obtained ?
Answer. (i) As seen from the graph, electrolyte A is a strong electrolyte which is completely ionised in solution.With dilution, the ions are far apart from each other and hence the molar conductivity increases.
(ii) To determine the value of limiting molar conductivity for electrolyte B, indirect method based upon Kohlrausch law of independent migration of ions is used.
Question. State Kohlrausch law of independent migration of ions. Why does the conductivity of a solution decrease with dilution ?
Answer. Kohlrausch law of independent migration of ions :The law states that limiting molar conductivity of an electrolyte can be represented by the sum of the individual contributions of the anion and cation of the electrolyte.
Λ° = v+Λ°+ + v–Λ°–
On dilution, the conductivity (k) of the electrolyte decreases as the number of ions per unit volume of solution decreases.
Long Answer Type Questions-I
Question. When a certain conductance cell was filled with 0.1 M KCl, it has a resistance of 85 ohm at 25°C. When the same cell was filled with an aqueous solution of 0.052 M unknown electrolyte, the resistance was 96 ohms. Calculate the molar conductance of the
electrolyte at this concentration.
[Specific conductance of 0.1 M KCl = 1.29 × 10–2ohm–1 cm–1]
Answer. Calculation of cell constant
Conductivity of 0.1 M KCl = 1.29 × 10–2 Ω–1 cm–1
Resistance = 85 ohm
Conductivity,
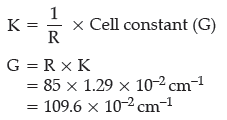

Question. The electrical resistance of a column of 0.05 M KOH solution of diameter 1 cm and length 45.5 cm is 4.55 × 103 ohm. Calculate its molar conductivity.
Answer. A = πr2
= 3.14 ´ 0.5 ´ 0.5 cm2
= 0.785 cm2
l = 45.5 cm
G* = I/A = 45.5 cm/0.785 cm2
= 57.96 cm-1
k = G*/R
= 57.96 cm-1/4.55 x 103 Ω = 1.27 ´ 10-2 S cm-1
Λm = k ´ 1000/C
= [1.27 ´ 10-2 S cm-1] ´ 1000/0.05 mol/cm3
= 254.77 S cm2 mol-1
Question. (i) State the law which helps to determine the limiting molar conductivity of weak electrolyte.
(ii) Calculate limiting molar conductivity of CaSO4 (limiting molar conductivity of calcium and sulphate ions are 119.0 and 160.0 S cm2 mol–1 respectively)
Answer. Kohlrausch law of independent migration of ions :
(i) The limiting molar conductivity of an electrolyte can be represented as the sum of the individual contribution of the anions and cations of the electrolyte.
(ii) Λ°m(CaSO4) = LoCa2+ + Λ° SO4 2-
= 119.0 S cm2 mol–1 + 160.0 S cm2 mol–1
= 279.0 S cm2 mol–1
Question. Calculate the molar conductivity and degree of dissociation.
Conductivity of 2.5 × 10–4 M methanoic acid is 5.25 × 10–5 S cm–1.
Given : Λ°(H+) = 349.5 S cm2 mol–1 and Lo (HCOO–)
= 50.5 S cm2 mol–1.
Answer.

Topic-2
Redox Reaction, Electrochemical Cell, Galvanic Cell,EMF of a Cell, Standard Electrode Potential, Nernst Equation
Very Short Answer-Objective Type Questions
A. Multiple Choice Questions:
Question. Electrode potential for Mg electrode varies according to the equation :
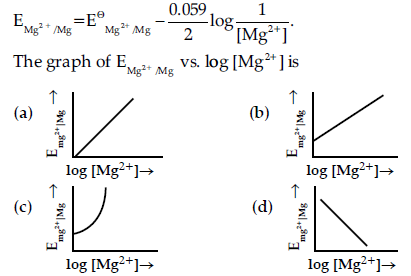
Answer
B
Question. In the electrolysis of aqueous sodium chloride solution which of the half cell reaction will occur at anode?
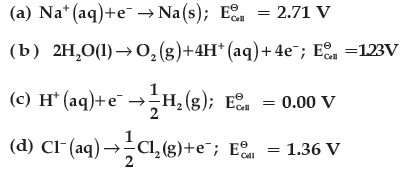
Answer
B
Question. The difference between the electrode potentials of two electrodes when no current is drawn through the cell is called ___________.
(a) Cell potential
(b) Cell emf
(c) Potential difference
(d) Cell voltage
Answer
B
Question. Using the data given below find strongest reduction agent.

Answer
B
Question. Which cell will measure standard electrode potential of copper electrode?

Answer
C
C. Answer the following:
Question. Depict the galvanic cell in which the cell reaction is Cu + 2Ag+→ 2Ag + Cu2+.
Answer. Cu|Cu2+|| Ag+|Ag
Question. Under what condition is ECell = 0 or ΔrG=0?
Answer. At equilibrium, that is, when the cell is completely discharged.
Question. Consider the following diagram in which an electrochemical cell is coupled to an electrolytic cell. What will be the polarity of electrodes ‘A’ and ‘B’ in the electrolytic cell?
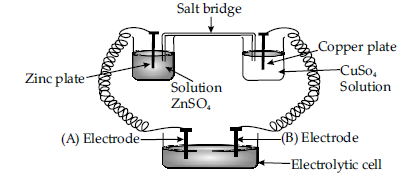
Answer. ‘A’ will have negative polarity; ‘B’ will have positive polarity.
Short Answer Type Questions
Question. The standard electrode potential (E°) for Daniell is +1.1 V. Calculate the ΔG° for the reaction.
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s) (1 F = 96500 C mol–1)
Answer. ΔG° = – n FE°cell
= – 2 × 96500 C mol–1 × 1.1V
= – 212300 J mol–1
or = – 212.3 kJ mol–1
Question. Suggest a list of metals that are extracted electrolytically.
Answer. Metals that are on the top of the reactivity series such as sodium, potassium, calcium, lithium, magnesium, aluminium are extracted electrolytically.
Question. Following reactions can occur at cathode during the electrolysis of aqueous silver nitrate solution using Pt electrodes:
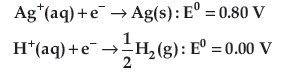
On the basis of their standard electrode potential values, which reaction is feasible at cathode and why?
Answer.
Ag+(aq)+e−→ Ag(s)
Because it has higher reduction potential.
Question. (i) Following reactions occur at cathode during the electrolysis of aqueous silver chloride solution :
Ag+(aq) + e– → Ag(s) E° = +0.80 V
H+(aq) + e– → 1/2 H2(g)
E° = 0.00 V
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
(ii) Define limiting molar conductivity. Why conductivity of an electrolyte solution decreases with the decrease in concentration ?
Answer.
(i) Ag+(aq) + e– → Ag(s); E° = + 0.80 V.
H+(aq) + e– → 1/2 H2(g) ; E° = 0.00 V.
On the basis of their standard reduction potential (E°) values, cathode reaction is given by the one with higher E° values.
Thus Ag+(aq) + e– → Ag(s) reaction will be more feasible at cathode.
(ii) Limiting molar conductivity : When the concentration approaches zero, the molar conductivity is known as limiting molar conductivity.
It is represented by Λ°m.
The conductivity decreases with decrease in concentration due to decrease in the no. of ions that carry the current in a solution.
Question. (i) On the basis of the standard electrode potential values stated for acid solutions, predict whether
Ti4+ species may be used to oxidise Fe(II) to Fe(III)
Ti4+ + e– → Ti3+ E° = +0.01V
Fe3+ + e– → Fe2+ E° = +0.77V
(ii) Based on the data arrange Fe2+, Mn2+ and Cr2+ in the increasing order of stability of +2 oxidation state. (Give a brief reason)
E°Cr3+/Cr2+ = –0.4V
E°Mn3+/Mn2+ = +1.5V
E°Fe3+/Fe2+ = +0.8V
Answer. (i) Since Ti4+/Ti3+ has lower reduction potential than Fe3+/Fe2+, it cannot be reduced in comparison with Fe3+/Fe2+ ions.
Hence Ti4+ cannot oxidise Fe2+ to Fe3+.
As the value of reduction potential increases the stability of +2 oxidation increases.
Therefore correct order of stability is Cr3+/Cr2+ < Fe3+/Fe2+ < Mn3+/Mn2+
Question. In a galvanic cell, the following cell reactions occurs:
Zn(s) + 2Ag+(aq) → Zn2+(aq) + 2Ag(s)
E0cell = +1.56 V
(i) Is the direction of flow of electrons from zinc to silver or silver to zinc?
(ii) How will concentration of Zn2+ ions and Ag+ ions be affected when the cell functions?
Answer. (i) Zinc to silver
(ii) Concentration of Zn2+ ions will increase and Ag+ ions will decrease.
Question. Calculate the emf of the following cell at 298 K Cr(s)/Cr3+ (0.1M)//Fe2+ (0.01M)/Fe(s)
[Given: E°cell = + 0.30 V]
OR
The conductivity of 10-3 mol/L acetic acid at 25°C is 4.1 × 10–5 S cm–1. Calculate its degree of dissociation if L0m for acetic acid at 25°C is 390.5 S cm2 mol-1.
Answer. 2Cr(s) + 3 Fe2+(aq.) → 3Fe(s) + 2Cr3+ (aq.)
n = 6

Long Answer Type Questions-I
Question. 6. A galvanic cell consists of a metallic zinc plate immersed in 0.1 M Zn(NO3)2 solution and metallic plate of lead in 0.02 M Pb(NO3)2 solution. Calculate the emf of the cell. Write the chemical equation for the electrode reactions and represent the cell.
(Given : E°Zn2+/Zn = – 0.76 V; E°Pb2+/Pb = – 0.13V)
Answer. Anode reaction : Zn(s) → Zn2+(aq) + 2e–
Cathode reaction : Pb2+(aq) + 2e– → Pb(s)
Cell representation :
Zn(s)/Zn2+(aq)||Pb2+(aq)/Pb(s)
According to Nernst equation :
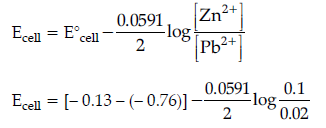
= 0.63 – 0.02955 × log 5
= 0.63 – 0.02955 × 0.6990
= 0.63 – 0.0206 = 0.6094 V
Question. Calculate ΔrG0 and log Kc for the following reaction at 298 K.
2Cr(s)+3Fe2+(aq)→2Cr3+(aq)+3Fe(s)
[(E°cell = 0.30 V), IF = 96500C mol-1]
Answer. ΔrG0 = -nFE°cell, n = 6
= -6 × 96500 C/mol × 0.30 V
= -173700 J/mol = -173.7 kJ/mol
E°cell = 0.059V/n × log Kc
log Kc = 0.30 V × 6/0.059V = 30.5
Question. Calculate e.m.f. of the following cell at 298 K :
2Cr(s) + 3Fe2+ (0.1M) → 2Cr3+ (0.01M) + 3Fe(s)
E°(Cr3+ | Cr) = – 0.74
E° (Fe2+ | Fe) = – 0.44 V.
Answer. E°cell = E°cathode – E°anode
= (– 0.44) – (– 0.74) V ½
= 0.30 V

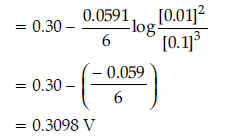
Question. Consider the following reaction :
Cu(s) + 2Ag+(aq) → 2Ag(s) + Cu2+(aq)
(i) Depict the galvanic cell in which the given reaction takes place.
(ii) Give the direction of flow of current.
(iii) Write the half-cell reactions taking place at cathode and anode.
Answer. (i) Cu(s) / Cu2+(aq) ll Ag+ (aq) / Ag(s)
(ii) Current will flow from silver to copper electrode in the external circuit.
(iii) Cathode: 2Ag+(aq) + 2e– → 2Ag(s)
Anode: Cu(s) → Cu2+ (aq) + 2e–
Question. Calculate the emf of the following cell at 25° C :
Fe | Fe2+ (0.001 M) || H+ (0.01 M) | H2(g) (1bar) | Pt(s)
E° (Fe2+ | Fe) = – 0.44 V E° (H+ | H2) = 0.00 V
Answer. Cell reaction is
Fe(s) + 2H+(aq) → Fe2+(aq) + H2(g)
E°cell = 0.00 – (– 0.44) = 0.44 V
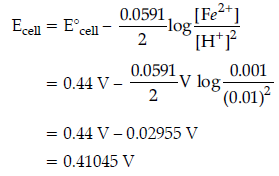
Question. (i) Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The limiting molar conductivity of ‘B’ increases 1.5 times while that of ‘A’ increases 25 times. Which of the two is a strong electrolyte?
Justify your answer.
(ii) The products of electrolysis of aqueous NaCl at the respective electrodes are:
Cathode: H2
Anode: Cl2 and not O2. Explain.
Answer. (i) ‘B’ is a strong electrolyte.
A strong electrolyte is already dissociated into ions,but on dilution interionic forces are overcome,ions are free to move. So there is slight increase in molar conductivity on dilution.
(ii) On anode water should get oxidised in preference to Cl-, but due to overvoltage/overpotential Cl- is oxidised in preference to water.
Question. A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal is placed in a one molar solution of AgNO3. An electrochemical cell is created when the two solution are connected by a salt bridge and the two strips are connected by wires to a voltmeter.
(i) Write the balanced equation for the overall reaction occurring in the cell and calculate the cell potential.
(ii) Calculate the cell potential, E at 25° C for the cell, if the initial concentration of Ni(NO3)2 is 0.100 molar and the initial concentration of AgNO3 is 1.00 molar.
[ E0Ni2+ /Ni = – 0.25 V, E0Ag+/Ag = 0.80 V]
log 10–1 = – 1 A
Answer.
(i) Ni(s) → Ni2+(aq) + 2e–
2Ag+(aq) + 2e– → 2 Ag(s)
Ni(s) + 2 Ag+(aq) → Ni2+(aq) + 2Ag(s)
E°cell = E°cathode– E°anode
= 0.80 – (– 0.25) = 1.05 V
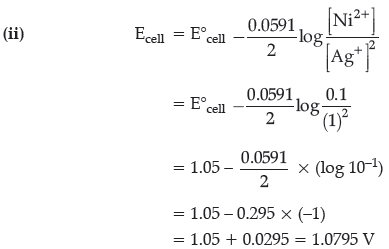
Long Answer Type Questions-II
Question. A voltaic cell is set up at 25°C with the half cells
Ag+ (0.001 M) Ag and Cu2+ (0.10 M) Cu. What
should be its cell potential ? [E° = 0.46 V, log 105 = 5]
Answer. Cu + 2Ag+ → Cu2+ + 2Ag
Half cell reactions :
Cathode (reduction) :
2Ag+ (0.001 M) + 2e– → 2Ag(s)
Anode (oxidation) :
Cu(s) → Cu2+ (0.10 M) + 2e–
n = 2
Eocell = 0.46 V

Question. (i) What is limiting molar conductivity? Why there is steep rise in the molar conductivity of weak electrolyte on dilution?
(ii) Calculate the emf of the following cell at 298 K:
Mg(s)|Mg2+ (0.1 M)||Cu2+ (1.0 × 10-3 M)|Cu(s)
[Given = E°Cell = 2.71 V]
Answer. (i) When concentration approaches zero, the molar conductivity is known as limiting molar conductivity.
The change in Lm with dilution is due to the increase in the degree of dissociation and consequently the number of ions in the total volume of the solution that contains 1 mol of electrolyte, hence Lm increases steeply.
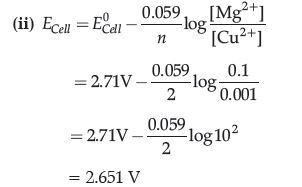
Question. Consider the given below figure (inside) and answer the following questions :
(a) Cell ‘A’ has Ecell=2V and Cell ‘B’ has Ecell=1.1V
which of the two cells ‘A’ or ‘B’ will act as electrolytic cell. Which electrode reactions will occur in this cell?
(b) If cell ‘A’ has Ecell = 0.5 V and Cell ‘B’ has Ecell = 1.1 V then what will be the reactions at anode and cathode?
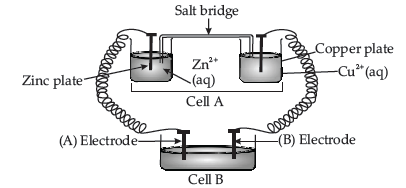
Answer. (a) Cell ‘B’ will act as electrolytic cell as it has lower EMF
Therefore, the electrode reactions will be :
Zn2+ + 2e– → Zn at cathode
Cu → Cu2+ + 2e– at anode.
(b) Now cell ‘B’ acts as galvanic cell as it has higher EMF and will push electrons into cell ‘A’.
The electrode reaction will be :
At anode : Zn → Zn2+ + 2e–
At cathode : Cu2+ + 2e– → Cu
Question. (i) Calculate E°cell for the following reaction at 298K :
4Al(s) + 3Cu2+ (0.01M) → 2Al3+ (0.01M) + 3Cu(s)
Given : Ecell = 1.98 V
(ii) Using the E° values of A and B, predict which is better for coating the surface of iron [E°(Fe2+/Fe)= – 0.44V] to prevent corrosion and why ?
Given : E° (A2+/A) = – 2.37V : E° (B2+/B) = – 0.14V A
OR
(i) The conductivity of 0.001 mol L–1 solution of CH3COOH is 3.905 × 10–5 S cm–1. Calculate its molar conductivity and degree of dissociation (a).
Given L° (H+) = 349.6 S cm2 mol–1 and Λ°
(CH3COO–) = 40.9 S cm2 mol–1
(ii) Define electrochemical cell. What happens if external potential applied becomes greater than E°cell of electrochemical cell ?
Answer. (i) Al(s) | Al3+(aq) (0.01M) || Cu2+(aq) (0.01M)|Cu(s)
LHE [Al(s) → Al3+(aq) + 3e–)] × 2 (Oxidation at anode)
RHE [Cu2+(aq) + 2e– → Cu(s)] × 3 (Reduction at cathode)
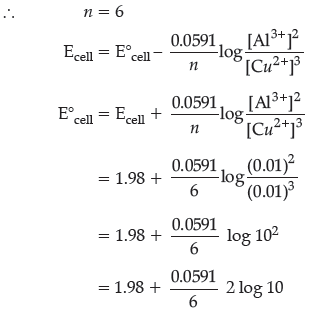

(ii) A is better than B because its E° value is more negative.
OR
i) C = 0.001 mol L–1, k = 3.905 × 10–5 S cm–1
Molar conductivity
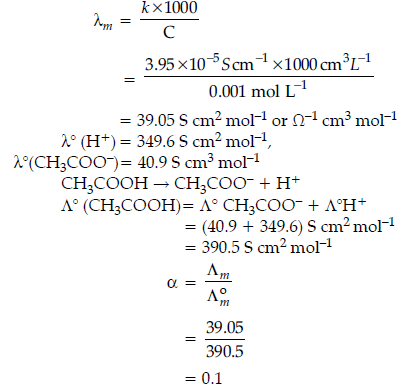
(ii) Electrochemical cell is a device used for the production of electricity from energy released during spontaneous chemical reaction and use electrical energy to bring about the chemical change.
If the external potential applied becomes greater than E°cell of electrochemical cell, the reaction gets reversed. It starts acting as an electrolytic cell and vice-versa.
Topic-3
Electrolysis, Laws of Electrolysis, Batteries, Fuel Cells and Corrosion
Very Short Answer-Objective Type Questions
A. Multiple choice Questions:
Question. While charging the lead storage battery________.
(a) PbSO4 anode is reduced to Pb.
(b) PbSO4 cathode is reduced to Pb.
(c) PbSO4 cathode is oxidised to Pb.
(d) PbSO4 anode is oxidised to PbO2
Answer. Correct option : (a)
B. Match of the following :
Question. Match the items of Column I and Column II.
Column I Column II
(i) Lead storage battery (a) maximum efficiency
(ii) Mercury cell (b) prevented by galvanisation
(iii) Fuel cell (c) gives steady potential
(iv) Rusting (d) Pb is anode, PbO2 is cathode
Answer. (i) → (d)
(ii) → (c)
(iii) → (a)
(iv) → (b)
C. Answer the following:
Question. Suggest two materials other than hydrogen that can be used as fuels in fuel cells.
Answer. Methane and methanol can be used as fuels in fuel cells.
Question. What are secondary cell?
Answer. Those cell which are rechargeable, i.e., in which products can convert back into reactants.
Short Answer Type Questions
Question. From the given cells :
Lead storage cell, Mercury cell, Fuel cell and Dry cell.
Answer the following :
(i) Which cell is used in hearing aids ?
(ii) Which cell was used in Apollo Space Programme ?
(iii) Which cell is used in automobiles and inverters ?
(iv) Which cell does not have long life ?
Answer. (i) Mercury cell,
(ii) Fuel cell,
(iii) Lead storage cell,
(iv) Dry cell.
Question. Write the name of the cell which is generally used in transistors. Write the reactions taking place at the anode and the cathode of this cell.
Answer. Dry cell/Leclanche cell
Anode: Zn(s) → Zn2+ + 2e–
Cathode: MnO2 + NH4+ + e– → MnO(OH) +NH3
Question. (i) Calculate DrG° for the reaction Mg(s) + Cu2+(aq) → Mg2+(aq) + Cu(s)
Given : E0cell = + 2.7 V, 1 F = 96500 C mol–1
(ii) Name the type of cell which was used in Apollo space programme for providing electrical power.
Answer. (i) Given, E°Cell = +2.71V and F = 96500 C mol–1, n = 2 (from the given reaction)
ΔrG° = – n × F × E0cell
ΔrG° = – 2 × 96500C mol–1 × 2.71V
= – 523030 J/mol
or – 523.03 KJ / mol
(ii) Hydrogen – oxygen fuel Cell / fuel cell.
Question. Write the name of the cell which is generally used in hearing aids. Write the reactions taking place at the anode and the cathode of this cell.
Answer. Mercury cell.
Anode: Zn(Hg) + 2OH– → ZnO(s) + H2O + 2e–
Cathode: HgO + H2O + 2e– → Hg(l) + 2OH–
Question. A current of 1.50 A was passed through an electrolytic cell containing AgNO3 solution with inert electrodes. The weight of silver deposited was 1.50 g. How long did the current flow ? (Molar mass of Ag = 108 g mol–1, 1F = 96500 C mol–1).
Answer. Quantity of charge required to deposit 108 g of silver = 96500 C
Quantity of charge required to deposit 1.50 g of silver = (96500/108) × 1.50 = 1340.28 C
Time taken = Q/I = 1340.28/1.50 = 893.5s
Question. Write the name of the cell which is generally used in inverters. Write the reactions taking place at the anode and the cathode of this cell.
Answer. Lead storage battery
Anode: Pb(s) + SO42-(aq) → PbSO4(s) + 2e–
Cathode: PbO2 + SO42-(aq) + 4H++ 2e– → PbSO4(s) + 2H2O(l)
Long Answer Type Questions-I
Question. Write the reactions taking place at cathode and anode in lead storage battery when the battery is in use. What happens on charging the battery ?
Answer. The cell reactions when the battery is in use are
given below :
Anode :
Pb(s) + SO42–(aq) → PbSO4(s) + 2e–
Cathode :
PbO2(s) + SO42–(aq) + 4H+(aq) + 2e– → PbSO4(s) + 2H2O(l)
i.e., overall cell reaction consisting of cathode and anode reactions is :
Pb(s) + PbO2(s) + 2H2SO4(aq) → 2PbSO4(s) + 2H2O(l)
On charging the battery, the electrode reactions are reverse of those that occur during discharge.
Question. (i) What are fuel cells ? Explain the electrode reactions involved in the working of H2 – O2 fuel cell.
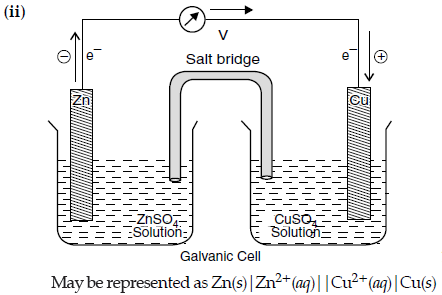
(ii) Represent the galvanic cell in which the reaction
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s) takes place.
Answer. (i) The cells which convert chemical energy of a fuel directly into electrical energy is known as fuel cells.
The electrode reactions are :
Anode :
[H2(g) + 2OH–(aq) → 2H2O(l) + 2e–] × 2
Cathode :
O2(g) + 2H2O(l) + 4e– → 4OH–(aq)
Net reaction :
2H2(g) + O2(g) → 2H2O(l)
Question. (i) Calculate the mass of Ag deposited at cathode when a current of 2 amperes was passed through a solution of AgNO3 for 15 minutes.
(Given: Molar mass of Ag = 108 g mol–1 1F = 96500 C mol–1)
(ii) Define fuel cell.
Answer. (i) m = Zit

= 2.01 g (or any other correct method)
(ii) Cells that converts the energy of combustion of fuels directly into electrical energy.
Question. (i) How many coulombs are required to reduce 1 mole Cr2O72– to Cr3+ ?
(ii) The conductivity of 0.001 M acetic acid is 4×10–5 S/cm. Calculate the dissociation constant of acetic acid if Λ°m for acetic acid is 390 S cm2 mol–1.
Answer. (i) Cr2O72– + 14 H+ + 6e– → 2Cr3+ + 7H2O
One mole Cr2O72– requires 6 moles of electrons for reduction.
Thus, quantity of electricity required
= 6 × 96,500
= 579000 Coulomb
= 5.79 × 105 Coulomb
(ii) C = 0.001 M
k = 4 × 10–5 S cm–1

Λm = 40 S cm2 mol–1
Λ° = 390 S cm2 mol–1
a = degree of dissociation of CH3COOH

Question. What is corrosion ? Explain the electro-chemical theory of rusting of iron and write the reactions involved in the rusting of iron.
Answer. The process of slowly eating away of the metal due to attack of the moisture and atmospheric gases on the surface of the metal resulting into the formation of compound such as oxides, sulphides, carbonates,sulphates etc., is called corrosion.
The electrochemical phenomenon of rusting of iron can be described as :
At anode : Fe(s) undergoes oxidation to releases electrons.
Fe(s) → Fe2+(aq) + 2e–
At cathode : O2(g) + 4H+ + 4e– → 2H2O(l)
Electrons released at anode move to another metal and reduce oxygen in presence of H+. It is available from H2CO3 formed from the dissolution of CO2 from air into water. H+ in water may be available also through dissolution of other acidic oxides from the atmosphere.
This site behaves as cathode
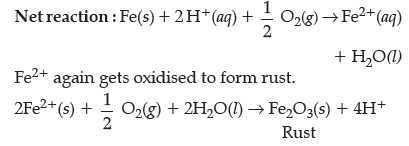
Question. (i) The cell in which the following reaction occurs :
2Fe3+(aq) + 2I–(aq) → 2Fe2+(aq) + I2(s)
has Eocell = 0.236 V at 298 K. Calculate the standard
Gibbs energy of the cell reaction. (Given: 1F = 96,500 C mol-1)
(ii) How many electrons flow through a metallic wire if a current of 0.5 A is passed for 2 hours? (Given:
1F = 96,500 C mol-1)
Answer. (i) ΔG° = -nFE°cell
n = 2
ΔG° = -2 × 96500 C/mol × 0.236 V
=-45548 J/mol
=-45.548 kJ/mol
(ii) Q = lt = 0.5 × 2 × 60 × 60
= 3600 C
96500 C = 6.023 × 1023 electrons
3600 C = 2.25 × 1022 electrons
Question. Silver is uniformly electro-deposited on a metallic vessel of surface area of 900 cm2 by passing a current of 0.5 ampere for 2 hours. Calculate the thickness of silver deposited.
[Given : the density of silver is 10.5 g cm–3 and atomic mass of Ag = 108 amu.]
Answer. Calculation of mass of Ag deposited :
The electrode reaction is Ag+ + e– → Ag
The quantity of electricity passed
= Current × Time
= 0.5 (amp.) × 2 × 60 × 60 (sec)
= 3600 C.
From the electrode reaction, it is clear that 96500 C of electricity deposit Ag = 108 g
3600 C of electricity will deposit Ag = (108/96500)× 3600
= 4.03 g
Calculation of thickness:
Let the thickness of silver deposited be x cm.
Mass = Volume × Density
= Area × Thickness × Density
(Volume = Area × thickness)
4.03 g = 900 (cm2) × x (cm) × 10.5 (g cm–3)
x = 4.03 cm (900×10.5) = 4.26 × 10– 4 cm.
Long Answer Type Questions-II
Question. (i) Define the following terms :
(a) Molar conductivity (Λm)
(b) Secondary batteries
(c) Fuel cell
(ii) State the following laws :
(a) Faraday first law of electrolysis
(b) Kohlrausch’s law of independent migration of ions
OR
(i) Define the term degree of dissociation. Write an expression that relates the molar conductivity of a weak electrolyte to its degree of dissociation.
(ii) For the cell reaction
Ni(s) | Ni2+(aq)|| Ag+(aq) | Ag(s)
Calculate the equilibrium constant at 25 °C.
How much maximum work would be obtained by operation of this cell ?
E°Ni2+/Ni= 0.25 V and E°Ag+/Ag= 0.80 V.
Answer. (i) (a) Molar conductivity of a solution at a given concentration is the conducting power of all the ions produced by 1 mol of an electrolyte.
(b) Secondary battery can be recharged by passing current through it in opposite direction so that it can be used again.
(c) Galvanic cells that are designed to convert the energy of combustion of fuels like hydrogen, methane, methanol, etc. directly into electrical energy are called fuel cells.
(ii) (a) Faraday’s first law of electrolysis states that the amount of chemical reaction which occurs at any electrode during electrolysis by current is proportional to the quantity of electricity passed through the electrolyte (solution or melt).
(b) According to Kohlrausch law of independent migration of ions limiting molar conductivity of an electrolyte can be represented as the sum of the individual contributions of the anion and cation of the electrolyte.
OR
(i) Degree of dissociation is the ratio of molar conductivity at a specific concentration to the molar conductivity at infinite solution.

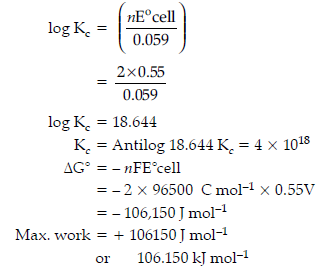
Question. (i) What are the two classifications of batteries ?
What is the difference between them ?
(ii) The resistance of 0.01 M NaCl solution at 25°C is 200 W. The cell constant of the conductivity cell is unity.
Calculate the molar conductivity of the solution.
OR
(i) What are fuel cells ? Give an example of a fuel cell.
(ii) Calculate the equilibrium constant (log Kc) and ΔrG° for the following reaction at 298 K.
Cu(s) + 2Ag+(aq) ⇌ Cu2+(aq) + 2Ag(s)
Given E°cell = 0.46 V and IF = 96500 C mol–1
Answer. (i) Classification of batteries :
(a) Primary batteries
(b) Secondary batteries
Primary batteries are non-chargeable batteries whereas secondary batteries are rechargeable.
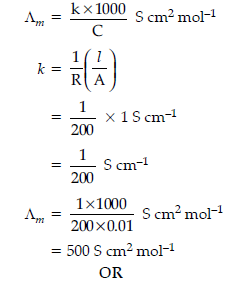
(i) Fuel cells are the cells which converts energy of combustion of fuel directly into electricity.
Example H2 – O2 fuel cell.

log Kc = 15.59
ΔrG° = – n FE°cell
= – 2 × 96500 C mol–1 × 0.46V
= – 88,780 J mol–1
= – 88.78 kJ mol–1
Question. (i) When a bright silver object is placed in the solution of gold chloride, it acquires a golden tinge but nothing happens when it is placed in a solution of copper chloride. Explain the behaviour of silver.

(ii) Consider the figure given above and answer the following questions :
(a) What is the direction of flow of electrons?
(b) Which is anode and which is cathode?
(c) What will happen if the salt bridge is removed?
(d) How will concentration of Zn2+ and Ag+ ions be affected when the cell functions?
(e) How will concentration of these ions be affected when the cell becomes dead?
Answer. (i) E˚ value of silver is lower than that of gold,hence silver displaces gold which gets deposited on the silver object.
E˚ value of copper is lower than that of silver,hence silver cannot displace copper from its solution.
(ii) (a) Electrons flow from Zn to Ag plate.
(b) Zn as anode and Ag acts as cathode
(c) Cell will stop functioning
(d) Concentration of Zn2+ ions will increase and that of Ag+ ions will decrease.
(e) No change
Question. (a) A cell is prepared by dipping a zinc rod in 1M zinc sulphate solution and a silver electrode in 1M silver nitrate solution. The standard electrode potential given:
E°Zn2+ / Zn = – 0.76 V, E°Ag+ / Ag = + 0.80 V
What is the effect of increase in concentration of Zn2+ on the Ecell ?
(b) Write the products of electrolysis of aqueous solution of NaCl with platinum electrodes.
(c) Calculate e.m.f. of the following cell at 298 K:
Ni(s) / Ni2+ (0.01 M) // Cu2+ (0.1M) / Cu (s)
[Given E°Ni2+/ Ni = – 0.25 V , E°Cu2+/Cu = + 0.34 V )
Write the overall cell reaction.
OR
(a) Apply Kohlrausch law of independent migration of ions, write the expression to determine the limiting molar conductivity of calcium chloride.
(b) Given are the conductivity and molar conductivity of NaCl solutions at 298K at different concentrations:
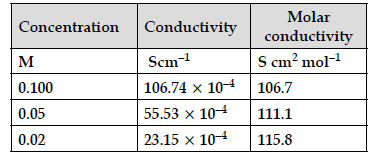
Compare the variation of conductivity and mola conductivity of NaCl solutions on dilution. Give reason.
(c) 0.1 M KCl solution offered a resistance of 100 ohms in a conductivity cell at 298 K. If the cell constant of the cell is 1.29 cm–1, calculate the molar conductivity of KCl solution.
Answer. (a) Ecell decreases.
(b) Anode: Cl2 ↑
Cathode : H2 ↑
(c) Cu2+(aq.) + Ni(s) → Ni2+(aq.) + Cu(s)
E°cell = E°cathode – E°anode
E°cell = 0.34 – (– 0.25)a
E°cell = 0.59 V

OR
(a) Λ°m(CaCl2) =λ°Ca2+ + 2λ°Cl–
(b) Conductivity of NaCl decreases on dilution as the number of ions per unit volume decreases.
Whereas molar conductivity of NaCl increases on dilution as on dilution the interionic interactions are overcome and ions are free to move.
(c) G* = kR

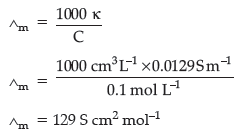
Question. (a) Write the cell reaction and calculate the e.m.f. of the following cell at 298 K :
Sn (s) | Sn2+ (0.004 M) || H+ (0.020 M) | H2 (g) (1 bar) | Pt (s) (Given : E°Sn2+/ Sn = – 0.14V)
(b) Give reasons :
(i) On the basis of E° values, O2 gas should be
liberated at anode but it is Cl2 gas which is
liberated in the electrolysis of aqueous NaCl.
(ii) Conductivity of CH3COOH decreases on dilution.
OR
(a) For the reaction
2AgCl (s) + H2 (g) (1 atm) → 2Ag(s)+2H+ (0.1 M)+2Cl–(0.1 M),
ΔG°= – 43600 J at 25°C.
Calculate the e.m.f. of the cell.
[log 10–n = –n]
(b) Define fuel cell and write its two advantages.
Answer. (a) Sn + 2H+ → Sn2+ + H2
(Equation must be balanced)
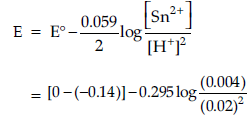
= 0.14 – 0.0295 log 10 = 0.11 V / 0.1105 V
(b) (i) Due to overpotential/ Overvoltage of O2
(ii) The number of ions per unit volume decreases.
OR
(a) ΔG° = – nFE°
– 43600 = – 2 × 96500 ×E°
E° = 0.226 V
E = E° – 0.059/2 log ( [H+]2[Cl–]2/ [H2] )
= 0.226 – 0.059/2 log[ (0.1)2 ×(0.1)2] / 1
= 0.226 -0.059 /2 log 10–4
= 0.226 + 0.118 = 0.344 V
(Deduct half mark if unit is wrong or not written)
(b) Cells that convert the energy of combustion of fuels (like hydrogen, methane, methanol, etc.) directly into electrical energy are called fuel cells.
Advantages : High efficiency, non polluting (or any other suitable advantage)
